[This article was substantially revised in February 2023, April 2019, and June 2019. Thanks to experience and advances in our software analytics, we’ve updated our approaches to sugars since this first went live in 2016.]
Sugars: they’re sweet, and they keep the ice cream soft. If you’ve had homemade ice cream with the consistency of concrete, it’s because the level of solids—especially sugars—was too low.
Some bloggers and cookbook authors tell you to soften the ice cream by adding alcohol. This works, but you can do better. While alcohol depresses the freezing point, it does so at the expense of smoothness. By increasing the unfrozen portion of water in the ice cream, while doing nothing to help control that water, it will encourage ice crystals to grow larger. You’ll end up with a softer but icier texture.
The Problem
The Solution
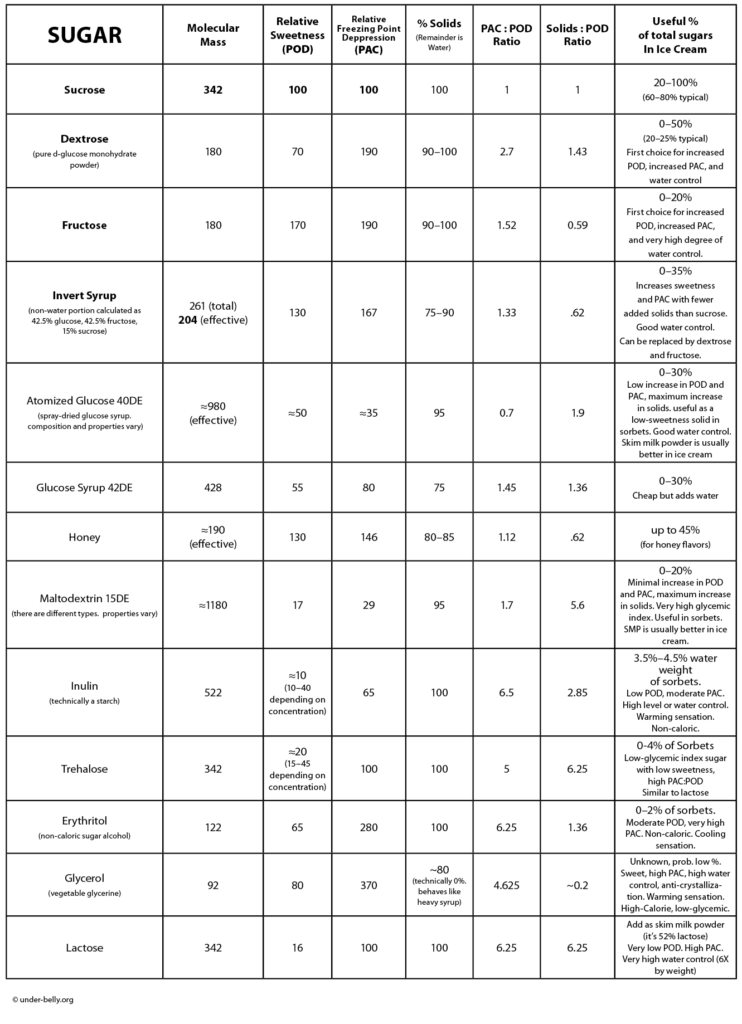
Dextrose (another name for the monosaccharide d-glucose) is about 3/4 as sweet as sucrose, but has nearly double the effect on freezing point suppression. Simply by decreasing sucrose and increasing dextrose, you can lower the sweetness while simultaneously softening the texture. Magic! Dextrose is also hygroscopic, meaning that it holds onto water, reducing the formation of both ice crystals and sugar crystals. It’s effectively a stabilizer, although it’s much less powerful than dedicated stabilizers.
Dextrose, fructose (and possibly invert syrup) can be especially helpful with flavors that require adding non-dairy fats, like cocoa butter (chocolate) and nut oils (nut butters). These fats tend to freeze harder milk fat, and give ice cream a dry, stiff, crumbly texture. Increasing the proportion of these monosaccharides can help preserve a smooth texture.
Other Important Structural Sugars
Atomized Glucose is useful in cases when you need maximum solids, high freezing point depression and minimum sweetness. It contains dextrose, and a whole stew of other mono-, di-, and polysaccharides, including dextrins. This gives it bulk without much sweetness. The trouble with atomized glucose is that it’s not a consistently defined product; you don’t know for sure what you’re getting or what its properties will be. The one clue manufacturers give us is the DE number (for Dextrose Equivalence). This tells us what percentage of the contents is reducing sugars (like dextrose) vs. larger saccharides (like maltodextrin). The higher the DE number, the more it will behave like dextrose powder. The lower the number, the more it will behave like maltodextrin. Lower numbers are more useful in ice cream; as a practical matter, most recipes use a mid-range number, like DE40, because this is most widely available.
Atomized glucose is a pantry staple for some pastry chefs, especially in Europe. It leads to some confusion, since some chefs don’t know the difference between it and dextrose, and some recipes don’t specify which to use. It also has the glycemic index (and associated health effects) of pure sugar. And its gotten expensive over the years. I avoid it except in sorbets and non-dairy ice creams, where its combination of solids and low sweetness can be ideal for creating a balanced formula.
Maltodexrin
Maltodextrin is a bit like atomized glucose minus the glucose. It’s got the same glycemic health concerns as atomized glucose. It adds solids and bulk with minimal effect on sweetness or freezing point. It’s a bit of an anti-sugar in this sense. It can be useful in particular sorbet flavors which by their nature are low on solids, and so need something to combat their innate wateriness—ones like lemon and watermelon. These flavors are built from fruit juices that are mostly water.It can also be useful in non-dairy ice creams. In ice cream, skim milk powder works better than maltodextrin. And the latter does all the bad things to your body that sugar does, without the benefit of tasting like anything. But milk solids are generally not an option in sorbets, which everyone expects to be dairy-free.
Inulin is technically a starch (derived from plants like chicory and Jerusalem artichokes), but we’re including it here because it offers some sugar-like behavior (some sweetness, some freezing point depression). It’s mostly used as a non-caloric fat replacer, since it has textural qualities similar to fats (this use is outside our purview; if you’re on some kind of diet, you’ve walked into the wrong room). It is, however, pure magic in sorbets, used at around 3.5% the water weight of the formula. It’s slightly exothermic, so it produces a mild warming effect when it melts in your mouth. This can counter the (usually undesirable) cooling effect of ingredients like erythritol. Inulin is interesting to us for cases where we need high solids without a lot of sweetness or freezing point depression. Nominally it doesn’t look as good as maltodextrin for this purpose, but inulin’s textural properties allow it to behave like a much larger dose of solids. You can think of it almost as a specialty stabilizer that also adds POD and PAC. As a bonus, it doesn’t have the lousy health impact of maltodextrin, and is non-dairy—which makes it an ideal choice for adding bulk and creaminess to sorbets.
Trehalose is a naturally occurring disaccharide composed of two glucose molecules stuck together. It’s much less sweet than sucrose, but has the same freezing point depression factor. Its properties are very close to those of lactose. But since the source of lactose is milk solids, it’s not useful in sorbets. Sorbets are the ideal application for trehalose, where it’s often a battle for getting the solids and freezing point correct without creating a cloying sugar-bomb. Trehalose does have a some disadvantages compared with lactose. It’s much less soluble, so we usually limit quantities to a few percent. Relatedly, it does not offer the incredible water control of lactose. It’s also an uncommon ingredient, in many cases leading us to special orders and jacked-up prices, so it’s good that we don’t need piles of it.
Erythritol is a naturally occurring sugar alcohol that provides sweetness with virtually no caloric value. We mammals don’t have the enzymes needed to break it down. Many sugar alcohols are, however, highly digestible to our gut bacteria, which can lead to gas and bloating. Erythritol is much less problematic in this regard. It’s primary drawback is its high price. It’s also endothermic, meaning that it that pulls in heat as it dissolves, creating a cooling effect in the mouth. This is refreshing in breath mints, but not so pleasing in an already-cold dessert. This can be combated with an exothermic ingredient (like inulin) or by just limiting the amount you use. The key benefit of erythritol is its very high PAC:POD ratio, making useful for controlling freezing point and sweetness in sorbets.
Lactose is the sugar that’s already in your milk and cream. Skim milk powder is about 50% lactose by weight—so you might as well just get your lactose from the SMP, which belongs in every recipe anyway. Lactose has the same freezing point depression factor as sucrose, but with sweetness that’s so low it’s barely detectable: highly useful for increasing solids and freezing point depression independently of sweetness. Lactose is also a champion in terms of water control. Lactose can absorb roughly six times its weight in water, which gives some stabilization power, and makes it more effective than most other sources of solids.
In Practice
[These guidelines differ somewhat from the formulas you’ll see elswhere in the blog series. We will eventually update those formulas to reflect this more refined approach]
How to Tweak:
Is the consistency ok but the sweetness too high? Less sucrose, more dextrose. Maintain PAC and decrease POD
Are you adding flavor ingredients that have their own sugars? Like fruit, chocolate, gianduja, or liqueur? Calculate (or estimate) the amount of added sugar and reduce the sucrose by the same amount.
With fruit, look up the actual composition of the fruit (it usually contains sucrose, fructose, glucose, and other sugars). You can compensate by reducing the glucose as well. We’ll discuss this in detail in a future post on fruit flavors. Or, as a shortcut, use calculated POD and PAC values for each fruit, and adjust the sugars to compensate.
More Sugars
Honey is a useful sugar in some ice cream flavors. It behaves mostly like invert syrup (because it IS mostly invert syrup—around 75% by weight), and tastes rather strongly … of honey. Because it adds about 20% water to the recipe, and increases body, it’s generally not a good idea to substitute honey for all the sucrose. But up to 50% works fine. It can be interesting to experiment with some of the more exotic and intense honey varieties, like buckwheat, heather, and chestnut. You’ll probably want to use these honeys in moderation. Mild honeys like clover and alfalfa are most traditional.
Atomized Glucose is a powdered sugar blend made by dehydrating glucose syrup, which itself my be derived from corn or wheat or other grains. Its contents and qualities vary widely from one product to another. The actual sugar content is going to be a blend of dextrose, maltodextrin, and a whole lot of other dextrins that fall between these two in terms of sweetness and other qualities. The one clue manufacturers give us is the DE number (for Dextrose Equivalence). This tells us what percentage of the contents is reducing sugars (like dextrose) vs. larger saccharides like maltodextrin. The higher the DE number, the more it will behave like dextrose powder. I like to avoid atomized glucose, because 1) you never know for sure what you’re getting, and 2) maltodextrin is a kind of phantom sugar; it has virtually no sweetness, but has the glycemic index (and associated health effects) of pure sugar. It also does nothing for flavor. It’s also gotten expensive over the years.
There ARE a few situations where atomized glucose is the best solution to a problem. In sorbets and dairy-free ice creams, it can provide the solids (and free water control) that you can’t get from milk solids. And it does it without excessive sweetness or freezing point depression. I try to use other options first, including trehalose, but often
Caramel is useful as a flavor ingredient. A little goes a long way, which is convenient—because it’s hard to know how caramel will effect the ice cream’s texture and freezing point. Caramelizing sugar is a gradual process by which some portion of the sucrose molecules break down into smaller molecules, and combine into larger, more complex, more flavorful ones. I like to use a small quantity of caramel, but to cook it to a fairly dark and flavorful degree. This way it will behave less like sugar in the recipe, and will have maximum effect on flavor.
You might also experiment with using caramels browned to different degrees—like a medium caramel, for more traditional toasted flavors, and a dark caramel, for the more complex and bitter burnt sugar flavors.
Caramel is challenging to calculate for. Its sweetness diminishes gradually as cooks darker, and at a certain point gets overtaken by bitterness. Its freezing point depression factor increases in the early stages of cooking, as the sucrose breaks down into monosaccharides. But as cooking progresses, larger molecules form, reducing the freezing point depression. Light-to-medium caramels can probably be treated like invert syrup.
Molasses is unrefined syrup centrifuged off from sugar cane syrup after it crystalizes. It contains all kinds of stuff, including water, so it’s best to use in small quantities just for flavor. The primary sugar component is sucrose.
Maple syrup is also useful as a flavoring. Like molasses, its primary sugar is sucrose (typically 52%), and it contains water (typically 45%) plus around 3% invert syrup.. It’s not easy to know precisely how much water is in there, since syrup is boiled down to whatever level the maker desires. Fortunately, a little goes a long way. Grade B is the most flavorful. The grade signifies darkness and not quality; annoyingly, many grocers don’t know their trade and stock only the inferior Grade A. It’s worth it to find a reliable local source of the good stuff. Maple syrup is so expensive these days, you should get all the flavor you can from every ounce.
There are other varieties of glucose, including corn syrups (typically around 1/3 glucose by weight) and various glucose syrups, identified by their DE number for dextrose equivalence. The DE number technically refers to the percentage of reducing sugars—in this case meaning either glucose or fructose. The higher the DE number of a glucose syrup, the more glucose it likely contains, and the greater the freezing point suppression. Atomized glucose is just spray-dried glucose syrup. It contains more water than anhydrous dextrose. Here’s all you need to know: Avoid using any of this stuff unless it’s all you can get your hands on. Pure Dextrose powder and invert syrup are more useful, and make it a lot easier to know what you’re getting. If you need a bulking ingredient, atomized glucose will do the job without adding water.
Non-Caloric Sweeteners
In the next post we’ll explore the dark arts of stabilizers.
Appendix 1: Invert Syrup
How to make Invert Syrup
Most professional kitchens just buy the stuff.
So—What is Invert Syrup?
Sucrose is a disaccharide, meaning a sugar molecule made up of two smaller monosaccharides: glucose and fructose. When we make invert syrup, we split these two monosaccharides apart, with the addition of water—a reaction called hydrolysis. Hydrolysis can occur with just the addition of water and heat, but an acid catalyst improves the efficiency of the reaction.
Typically, we can split (invert) about 85% of the sucrose. Manufacturers may be able to invert more of the sugar, by using other chemical or enzymatic catalysts.
When you cook your own, you control the final water content with the cooking temperature. Cooked to 113°C–114°C the final syrup will contain a bit under 20% water. This is dry enough to work in ice cream without adding too much water, and gives a long life in the fridge. But it’s not unreasonably gluey.
This inversion of polarized light has no known application in the kitchen. Not even Nathan Myhrvold has suggested that we run out and buy a polarimeter. Just try to remember that inverting sugar does not mean turning the bag upside-down.
Appendix 2: Sample Recipe
Quartet of Dark Sugars Ice Cream
I’ve written this for cooking in an immersion circulator, but it adapts fine to the stovetop or other heating methods.
25g maple syrup
10g (2 tsp) alcohol-based vanilla extract
-set immersion circulator to 75°C
-add yolks, cream and vanilla extract.
Milk Fat: 11.3%
Total Solids: 37.4% (a little lower than ideal)
Solids Nonfat: 25.3%
Milk Solids Nonfat: 10.6%
Acidity: 0.17%
Alcohol: 0.3%
Stabilizer/Emulsifier: 0.14%
Egg Lecithin: 0.29%
POD: 114 / 1000g
PAC: 263 / 1000g (a little higher than ideal)
Appendix 3: FPDF, PAC, Absolute PAC
Problems in Quantification






Hey, just found this blog, and spent most of the night pouring over it. Great job!
I found you because I'm on a search to better understand ice cream, specifically for the purpose of perfecting a sugar free version to fit into a ketogenic diet. There's nothing at all worthwhile to purchase commercially, nor can I find anything outside of mommy blogs and their "mason jar no churn ice cream"
I'm a chef myself, and have a professional kitchen and a lot of proper tools, so I want so be able to produce a professional grade product. Unfortunately I don't have the knowledge base to understand how to make a truly palatable texture without any sugar (including naturally occurring from dairy). I'm talking absolute minimal carbs, the lower the better, so I'd love to hear your thoughts.
Thanks for writing, chef.
I’m gradually learning a bit about sugar-free ice creams, largely because so many questions about them are coming in. Right I now I just have a few thoughts for you:
1. Some of the sugar alcohols, especially erythritol, make excellent substitutes because because they taste pretty close to sugar and have good freezing point depression. But you want to go easy, because too much can cause digestive distress (you don’t metabolize these ingredients, but your gut bacteria go to town on them).
2. Low-sugar ice creams (with much of the sugar replaced by sugar alcohols) are much easier to make well than no-sugar ice creams.
3. Make sure to get the total solids up to the right levels. Nonfat dry milk is your best friend.
4. Do a deep search on Pub-Med or Google Scholar, and let the scientists convince you to get off the ketogenic diet before you do yourself any harm!
Thanks for the quick reply!
Just a few follow-ups directly related to your answers:
1): erythritol is my go to sweetener, so that’s good news. I find I tolerate it quite well, so I’ll stick with it as a first choice. It’s not quite as sweet as sugar though, so how will the fact that I’m adding more volume to match sugar content affect the end product from a total solids point of view?
2) understandable, I’ll play around with minimum ratios. Dairy is already a big source of sugars, so if I’m going to try to make some sorbets or other non dairy options it may allow me to add back some sugar to adjust freezing points. Can you advise on which fats have the best (lowest) freezing temperatures so that I could increase the richness without dairy?
3)if I’m sticking with a dairy based ice cream, but don’t want to add milk powder because of the high sugar content, is there any other option that would work well to get total solids up without adding carbs in the form of either sugars or starches?
4) thanks for your concern on this. I’ve honestly spent the last 6 months trying to find a concrete, evidence based argument against the keto diet, as I was very skeptical at first.
Typically people argue that it’s not a magic pill, and that you can’t live on bacon and cheese. This is unfortunately what most people (including many that adhere to the diet) believe the diet consists of.
In reality,if you’re doing it correctly, you’re eating a wheelbarrow full of vegetables ever day, some lean, organic protein, and a good amount of healthy fats (not a bag of pork rinds).
It’s definitely not a diet for everyone, as it requires planning, some minor micronutrient supplementation, and at the end of the day is still bound to the calories in, calories out equation.
I just find it easier to stick within my caloric needs by eating this way, and I’m eating far more whole foods that I was before I started.
All this being said (sorry for the rant) I’m very interested in any sources you can point me towards that would show me reasons that I should reconsider the diet as a long term lifestyle. As I simply haven’t been able to find anything I’ve been satisfied with.
Hi Matt, sorry for the slow reply … your comment caught us right in the middle of transition from the old blog site, so it got delayed and also the formatting is little messed up. We’re trying to fix.
In the mean time …
1. erythritol’s challenge is that that it has relatively low sweetness (POD 0.65) and high freezing point depression (PAC 2.8). So at normal sweetness levels, you’ll tend to have very soft ice cream. This is one reason it’s often used with other sweetners.
2. and 3. You might consider inulin (technically a starch). It has fairly low sweetness, fairly low freezing point depression, but excellent fat-like textural properties. It behaves like a much larger quantity of solids. And is essentially non-caloric. Try it at 4-7%.
4. Concerns with the keto diet are that 1) it has poor long-term effectiveness, largely because the near-term gains are mostly from water loss and lean muscle mass loss. And 2) ketosis is an unhealthy metabolic state. Doctors and nutritionists prescribe it as a short-term, emergency intervention. It seems likely that most people on the keto diet aren’t really pushing themselves into ketosis, which makes that less of a concern. but it also raises questions about why they’re doing what they do.
I haven’t dug through the peer-reviewed research. Here’s a healthline article with a few citations: https://www.healthline.com/health-news/keto-diet-is-gaining-popularity-but-is-it-safe-121914#8
The final word on this topic is a long way off. Just be careful!
I'm not familiar with the AFP scale, so I'm not completely sure what the numbers mean. The scientific way to look at freezing point depression is simply to look at the molecular mass of the ingredient. The lower the mass, the greater the freezing point depression. And the relationship is linear.
Take a look at the table above. Invert syrup is listed as having a molecular mass of 204, which gives it 1.67 times the freezing point depression of sucrose (67% greater).
If AFP is based on sucrose, and if sucrose is 100, then it sounds like your gelato chef friend is giving you the numbers for either glucose or fructose; an understandable assumption, since invert syrup should just be a mix of these two. But because not all the sucrose gets inverted the math works out a bit differently.
Also keep in mind that any numbers given for invert syrup are estimates, because the actual water content and the actual efficiency of the hydrolysis can vary. Commercially available invert syrups will probably contain a lower percentage of residual sucrose than homemade versions, so their freezing point depression (and sweetness) may be a bit greater.
[Thanks to your question, I re-calculated the numbers for invert syrup in the chart. Previously I reported the total molecular mass, rather than giving a weighted average of the glucose, fructose, and residual sucrose. The new value is shown as the effective molecular mass.]
Hi Bruno,
yes, anhydrous glucose would be an excellent ingredient. I'm pretty sure that culinary dextrose is glucose monohydrate, which is equally good, and possibly cheaper. I like both of these much better than any glucose syrup.
I don't have data for 43DE glucose, but assuming it's similar to 42DE, it has about 60% the sweetness of dextrose, and 40% the freezing point depression. So I'd start by using dextrose / glucose powder at 50% the quantity of the syrup, by weight.
Hi,
I have question about Trimoline AFP.
What is correct AFP number in trimoline? I learn from Italian gelato chef he told me 190 AFP..
Its there has different trimoline?
Hi,
Compliments for the very informative blog.
My egg-less white mix recipe (about 6.5% fat) includes the following combination of sugars:
6% 43 BE GLUCOSE, 5% SUCROSE and 8% INVERTED SUGAR.
I’d love to substitute the annoying glue 43 BE glucose for a dry sugar equivalent.
Would anhydrous glucose powder be the best option? If so, what would be, let’s say, the equivalent for 1 kg of the 43 BE glucose.
Many thanks for your kind attention and sharing your knowledge.
Hi Anon,
yes, I've been playing with the spreadsheet a bit. I don't know my way around it well enough yet for it to be primary tool, but it's nicely done and I can see it becoming a big time saver. I may have to learn some more about Excel … there are some things I'd like to customize on it. Thanks again for sharing.
Did you ever get the spreadsheet working?
I've substituted fructose for invert syrup in several batches of ice cream now, and it works fine. I don't have the means to do side-by-side blind tastings, so don't take my word as gospel. But based on casual experience, and my understanding of the science, it doesn't make a difference—except that fructose allows you to add a bit less water to the recipe. Which would be a positive difference, if noticeable at all.
I find fructose easier to work with, and so will switch to it for my own use. But I'll keep posting recipes with invert syrup, since this is standard pastry kitchen ingredient.
The substitute blend I'm using in my standard ice cream base:
70g granulated sugar
30g dextrose
5g fructose
Hi Jonas,
to keep things simple I don't account for the water weight of the invert syrup. The stuff is usually around 20% water, we're talking about a few grams of water at most.
So with invert, honey, glucose syrup, I'm talking about the whole weight of the thing.
If you want your spreadsheet to be extremely precise, you could have it parse those values internally—so it would assume, for example, that 15g invert syrup means 6g glucose, 6g fructose, and 3g water). When I'm working things out by hand, this degree of precision gets unruly, but if a spreadsheet is doing the heavy lifting, why not?
It makes perfect sense that your alcohol experiment yielded an icy texture. Alcohol doesn't help control the unfrozen water, so you get softness at the expense of iciness. If you want to get really nitpicky, alcohol's will have a sweetness level that's somewhat higher than zero … although my guess is that you can ignore this. At the alcohol and sugar levels likely to be present in ice cream it's probably imperceptible.
Good luck with the spreadsheet. I'd love to see it.
Two Quick Questions:
"My starting point is 13% sugar by weight (not counting the lactose in the milk).
The blend consists of:
60% sucrose
26% dextrose
13% invert syrup"
Is the 13% inverted Syrup defined as "the weight of the contributing sugars (not including water)" or "the total weight of the inverted syrup (including water)"?
In addition, in your table, defining freezing points and sweetness factors, does this already account for the water content in inverted syrup or honey or is it just considering the sugar components of the ingredients?
I'm working on a spreadsheet that you can create ice cream recipes on and have it automatically generate it's ingredient breakdown (i.e water percentage, milk fat percentage, etc.). With that spreadsheet, I am working to account for different sugars. At the end of the spreadsheet, I have a sugar sweetness rating (Based on sweetness of the total sugars per total volume) and The sugar equivalent of freezing potential (total percentage of sugar at relative freezing suppression of 1.00). I have also accounted for alchohol (0 sweetness, 7 freezing suppression) but found that the mixture was icy . I just want to make sure I understand the information in this section so I am calculating the correct sugar amounts
Hi Jonas,
Another reply mostly for the benefit future readers. I’ve significantly updated the information on this page to reflect some new approaches. The ratios you’re asking about have been replaced by new ones.
Hi, is it possible to find thos excel file somewhere?
Thank you
Dave Arnold, former technology director at the International Culinary Center in NYC, has confirmed that powdered glucose and fructose should work the same as invert syrup.
Thanks for taking the time to discuss this, would you mind updating your blog with more information? It is extremely helpful for me.
Dry Ice Blasting in Dubai & Dry ice Dubai
Hi Jerome, thanks for writing. Dextrose and glucose are interchangeable names for the same sugar. The only likely difference in practice is that products labelled Dextrose are (usually) the anhydrous or monohydrated powdered forms of the pure sugar. These versions contain little or no water.
Sugar labelled Glucose, when sold in baking supply stores, is often either a syrup or "atomised glucose"—which is a powder made by spray-drying glucose syrup. Both of these products contain a lot more water than the Dextrose powder products. They also contain a range of other carbohydrates, including maltodextrin and other dextrins. You’ll never know exactly what the contents are. The only clue is from the DE (dextrose equivalence) number. Dextrose powder has a value of 100; glucose syrup and atomized glucose powder can range from DE-15 to DE-80 or so. The lower the DE value, the less sweet and the more it will behave like maltodextrin. The higher, the sweeter, and the more it will behave like dextrose.
It’s also possible that some products sold as Glucose are identical to Dextrose powder, but it can be hard to tell.So it makes life much simpler to just use Dextrose powder.
Thank you for sharing all these informations. However I'm wondering about the use of glucose, which is almost always used by professionnals, here in France.
In contrary, dextrose is not recommended by french ice cream makers because it might significantly limit overrun. What do you think about this ?
Anon, I'm so glad it worked out. That's one of my favorite recipes.
"Quartet" recipe came out GREAT! My wife said it would probably pair with a nice stout!
Boy, to think of all the failed batches I could have prevented eating if only I found this site first. So…many…calories…
Not only is this the best technique around it is all explained in ways that make sense. Excellent job, I just wish you were able to post more often! (but grateful to get it when you're able)
THANKS!!!
I asked chef Laiskonis your question. He doesn't see why it wouldn't work, and suggests trying it. The substitution isn't interesting to him, because as a pastry chef, he always has invert syrup on hand, but doesn't use fructose powder.
I find the idea compelling, though—because it's easier to work with powdered ingredients than with a heavy syrup in ice cream. I haven't tried it yet, because to to set up a controlled experiment is difficult, and I don't have facilities for making identical, simultaneous batches of ice cream.
I'm looking for a food scientist to ask.
Thanks for looking further into this.
Anecdotally, glucose has been working well in my home experiments.
I’m curious to know the answer to this as well. I can’t imagine how invert sugar wold impart any benefit aside from the freezing point depression of the inverted sugar components. Maybe the viscosity plays a role? Though I would think this would be lost when it’s heated and dissolved with the mix.
Please can you help me.i am putting 250 gr sugar,50 gr dextrose,50 gr fruit base in 1 litre.but i must use emulgator because my fruit ice cream is like sand.but the problem is i dont like using emulgator because its brightening my ice cream,what would i do ?i dont wont to use it
Hi Paul,
Sorry to take so long with a response. Your question has occurred to me before, and I've never been able to find an answer. There's no shortage of sources that go on endlessly about the benefits of invert syrup. None that I've found say why (or if) it's any different from a glucose and sucrose dissolved together.
I'm waiting for answers from a couple of real chemists and will let you know what i find out.
First of all, thank you for sharing your knowledge on ice cream! Your articles are a pleasure to read and have taught me a lot.
I have a quick question regarding invert sugar:
Are the stabilizing & textural benefits of invert syrup attributable to its monosaccharide constituents, or is there some other underlying cause?
Would I get the same effect by using a proportional amount of anhydrous glucose / fructose in place of invert syrup?
I think Lebowitz is just trying to keep things simple for people. Alcohol works. But unlike sugars, it doesn't do anything to improve the ice cream's texture. By lower the freezing point without adding anything to improve body or stability, alcohol actually results in an icier ice cream, not a smoother one.
In a later post I'll get into alcohol flavors—for these we add more nonfat milk solids, and change the sugar blend so we don't get too much softening.
David Lebowitz says use alcohol to soften ice cream. What's wrong with this?
Hello there, and thank you for these incredible articles.
Your description of the thyme ice cream by Bajolle has caught my imagination. Do you happen to know, or have an educated guess about the formula for it?
Thanks, T.M.
That was a long time ago, and I’m sure I’ve romanticized it. My best guess is that it was a fairly rich, custard-based ice cream, with the thyme steeped into the mix on the stovetop … something I might not find especially impressive today, but that was a revelation at the time.
Hi,
I have a question about the sugar content of fruits. Do you have a good resource for finding the fructose, glucose, and sucrose break down of each fruit? I find that assuming all the sugar is from sucrose gives too much variation in results. Some of my fruit ice creams and sorbets come out rock hard, while others come out perfect even though they have very similar sugar and total solids percentages.
Hi Meg,
you’re in luck. I just started the series on fruit flavors. Next post is going to include data tables.
Stay tuned!
It’s never going to be an exact science, because every batch of fruit will be different. Even if you test with a brix hydrometer, you’ll be working with approximations. But with a little effort you can be sure the results will always be good, even if not perfectly consistent.
Hello again,
I am wondering if you know of a good source where I can find the relevant ice cream chemical values for a range of ingredients. Most specifically I am interested in POD, PAC, PAC (sa), MSNF, SNF, Milk Fat, Fat. I am relatively new to the analysis of my ice cream along these lines. I generally operate off the “add more this” or “less that” in a very haphazard way. This is really helping me to quantify this learned experience and a strong resource (book, website, etc) would allow me to forecast results and waste less time when experimenting with new flavors. Thank you for your response and commitment to your blog.
Thanks for all the work that you do to present the science of ice cream making in a clear concise way. that being said what is a good sugar blend using erythritol that makes it somewhat healthier that sucrose and dextrose blend. I am a novice just follow recipes without much deviation but would like to understand the science to pursue my true passion granita. thanks
Invert sugar question.
Whatever I do I can’t get the calculations to fit your PAC and POD values.
I checked a commercial brand on amazon.
It had 18% water and 95% of the sugar was converted.
So I assume
Water 18%
Sucrose 4.1%
Fructose 38.95%
Glucose 38.95%
Then the PAC and POD for this by weight is PAC=152.1 and POD=97.6
Even when testing other scenarios with more residual sucrose etc I can’t get the POD up to 130.
When testing the values you have in your table using 15% sucrose and 42.5% fructose and 42.5% glucose and zero water I get PAC=176.5 and POD=117.
What am I missing?
15×100/100 + 42.5×74/100 + 42.5×173/100 = 120 for the POD . I think the ‘130’ value is some sort of mistake brought forward from book to book but never really checked. I only recently realised this myself… Also, it is formally incorrect to give a ‘total’ molecular mass for a mixture of ingredients, like invert sugar is.
You may be correct on this, although it’s important to note that the POD of a sugar can only be estimated mathematically. It’s an attempt to capture a rather complex sensory phenomenon in a single number. Variables include how quickly the sensation of sweetness builds (and dissipates), and how the sweetness tracks with temperature. It’s all determined by giving test subjects different concentrations of sugar water and comparing to some standard.
I usually avoid invert syrup, because you also never know how much water is in there, so it’s always a guessing game. And it’s a pain in the ass to work with. Pastry chefs like it because they always have it.
Also, I think the line on inulin is not completely correct, if I may add my two cents.
Firstly, inulin is not a starch (a digestible – by humans – glucose polymer) but a soluble fibre (digestible by our gut bacteria) and it’s a polymer of fructose molecules. This is the reason why some people cannot tolerate it – it may cause bloating and even diarrhoea.
Secondly, as there are various types of inulin, their Degree of Polymerisation clearly has relevance both on freezing point depression and on sweetness. In general, for chicory inulin, the Sucrose Equivalence is considered to be about 25, at least the one we get to buy in Italy to make our gelato, so the average molecular weight of the chains is higher.
Having said that, I agree that inulin works wonders in sorbets and if you can tolerate it is a good addition in any case where you may lack solids, especially if you are not using MSNF
I am working on sugar free (no added sugar) icecream . Tried out with erythritol in my regular base instead of sugar, since it has a high PAC, and that was just what I wanted.
It was fine in texture when I got it out of the ice machine – but after freezing in my storage freezer it was as hard as rock – and erythritol was really giving an odd taste.
How can I overcome the issue?
Wow great post! I’ve learned SO much! That chart is incredibly useful. I’ve grown to love glucose syrup (no atomized where I live). It’s a perfect way to reduce sweetness and let flavors shine. I just made a vegan peanut butter Oreo ice cream with your vegan recipe (slightly modified of course) and the flavor came out beautifully! It was so flavorful that it made me realize just how crappy every other ice cream I’ve ever had is. I think relying on sweetness is lazy and creates a cheap flavor. The only downside is that those.on a budget like me can’t use cheap crappy favor ingredients lol but it’s worth it!
I’ve learned so much about the science of ice cream from this site, thanks for all your work in compiling this info and explaining it well. I have been using invert syrup in many things besides ice cream as well. I had a fun idea: can you make a similar ‘invert’ syrup from lactose, splitting it into the component glucose and galactose parts? In theory, it would have the same PAC as the sucrose invert syrup, but would be around 42% as sweet, based on my calculations for an 80% syrup that is 85% inverted (so, 20% water, 34% glucose, 34% galactose, and 15% lactose). Didn’t know if you knew of anyone trying it or any reason it shouldn’t work. Cheers!
Great question, and I have no idea. I haven’t seen any literature on sugars besides sucrose being “inverted,” possibly because a glucose/fructose syrup has special qualities that are useful in pastry and ice cream.
I have no idea what properties you’d get if you broke lactose down to its constituent saccharides, how they’d function in ice cream, or how you’d do it (since we don’t generally add lactose as an ingredient; it’s bound up with the dairy products.
Good question for someone like Goff or Cesar Vega.
Great article!! I’m trying to make a creamy fudgesicle with 10+ g of protein and 140-180 calories. I am using protein powder and Greek yogurt for sure.
I’ve experimented with honey, stevia, maple syrup and monk fruit. Close but no home runs.
Any tips? They all taste fine but they separate when frozen and often are icy
I’m somewhat surprised that Xylithol wasn’t mentioned… is there a reason for that?
(looks like ice creamer Denada swears by it)
Hey,
Such a great blog! You’ve effectively saved me years of trial and error experimentation.
I’m lucky enough to own a conventional ice cream churner (lusso) along with a Pacojet and a Ninja. I have been on a mission for the last six months or so to make the best possible ice cream with low fat and low sugar.
Solids are a major issue when making these ice creams, PAC and POD and balancing is difficult to get right. What I have so far (for a basic vanilla base) is:
1. Erythritol is my main bulk sweetener, and it works well when using the Lusso and the Ninja. The Pacojet however seems to impart too much friction heat and thus yields a far too soft ice cream. I much prefer the Pacojet in terms of texture and convenience (the Ninja is not bad but it has some serious drawbacks) so I am looking to reduce PAC while maintaining or even increasing POD. The erythritol is the main culprit in terms of high PAC. So instinctively I think I should try some more intense sweetener while pulling back erythritol. Stevia perhaps? Any thoughts?
2. Fat replacers. Generally a Pacojet seems to work best with fat levels at the lower end – which is good news for me as I want to keep fats at a minimum. No idea how that translates to fat replacers but it seems to hold true. I am using Inulin at around 6% currently.
3. Milk and milk solids. I am using 3% lactose free milk currently. Replacing milk powder I am using a blend of sodium caseinate and whey protein. Not obligatory but I would also like to incorporate a high level of protein which is why I am using these. I am thinking sweetness from lactose specifically isn’t required.
4. Vanilla is real, no extracts as I don’t want to add alcohol for fear of PAC issues.
5. Thickeners, stabilizers and emulsifiers. Guar, CMC, Poly 80 and Mono di glycerides. This part is hardly scientific but rather inspired by Google searches.
My issue with above is that I am not hitting levels of solids above 25-30%. 30% is not ideal in terms of nutritional values (KCAL) and PAC. Inulin can compensate a bit from my understanding but my Ice cream is still coming out softer than I would like it. I haven’t tried Stevia yet, but will as the sweetness is muted using only Erythritol. Texture is what is most lacking but likely the easiest to sort out. Latest batch came out thick and a bit gummy. I will adjust the hydrocolloids.
Many questions but the main one is: Any magic bullet in terms of PAC while maintaining a very lean low carb ice cream?
Latest batch was:
450 grams of lactose free 3% milk
30 grams erythritol
1 gram of stevia
35 grams of inulin
20 grams of whey
20 grams of sodium caseinate
1 gram salt
2 grams vanilla
1 gram each guar, cmc, poly 80 and mono di glycerides
This gave me a POD of 110, total PAC of around 200
I would love to hear your thoughts on the matter.
Edit to above.
The mix is frozen to -23/24 C. The Ninja and Pacojet both warm it up by 14-16 C during processing. In other words I need to optimize for a serving temperature of approx -6/7/8 C. Above batch is not far off though it is still too soft, and the solids are only around 27%. The gold standard for me is the Modernist Cuisine Pistacchio Gelato (made without dairy). It is super well balanced for a Pacojet. Total PAC 250 and POD 150. However it packs a massive caloric punch, which is what I want to eliminate.
This was such a good read, and I’m so surprised I haven’t heard of dana cree until today, as i’ve worked at both alinea and the publican! Great post, i’ve been working on some new ice creams for the restaurant and this is really inspiring me to take it to the next level.