How Much Liquor?
For weaker spirits, like wine, the problem isn’t the added alcohol as much as the added water. You can reduce a portion of the wine with heat, but not most of it; heat will kill the aromatic flavors. I find it best to replace a portion of the milk with the wine—then add a commensurate amount of nonfat dry milk, to make up for the lost solids.
Ultra boozy ice cream is becoming a trend, with companies like Tipsy Scoop churning out 5% alcohol concoctions. When you get into this range, it’s more about a sweet frozen cocktail than about a liquor-flavored ice cream. Getting you buzzed takes a higher priority than texture or flavor. And you have to start carding people. If you want to head in this direction, just follow the instruction here, but take everything farther.
Liquor Quality
For example, with cognac, I use an affordable VSOP grade that I’d be happy to drink, but that I wouldn’t be tempted to ruminate over with my pompous friends. I like Gosling’s rum—bold, full-flavored, if not the most complex. Save the really good / delicate stuff for drinking straight.
Summary of basic compensations:
A Guide for Wine Ice Creams (this is a work in progress)
If you experiment with this template, please let me know how it goes.
Thoughts on Cocktail Flavors
Generally, cocktails that are already sweet (anything from variations on the Old Fashioned to Tiki drinks) will work better than dry cocktails. I don’t see much potential for Martini ice cream (but maybe as a sorbet …)
Obviously, leave out the simple syrup. And avoid vodka cocktails (see above. And they’re not real cocktails).
Go to good cocktail bars. Figure out what the mixologists are doing. The best of these guys are essentially chefs. Notice how they layer and balance the flavors. Notice how in some cases all the individual flavors remain identifiable; in other cases, an entirely new flavor is created by the combinations. Try to figure out what’s going on. Look at the structure of classic cocktails (and the often similar modern ones). Try to accomplish the same effects in the flavors you create.
Sample Recipe (single spirit)
-harden for several hours (preferably overnight) in a cold freezer. freezer should be set to -5°F / -20°C or lower. Ice cream will have to warm up several degrees before serving. 20 to 30 minutes in the fridge works well. Ideal serving temperature is 6 to 10° F / -14 to -12°C.
Sample Recipe 2 (cocktail)
The Bourbon Smash is one of my favorite summer cocktails. It would be one of my favorite winter cocktails, too, but it can be hard to find fresh mint outside the summer months. I just made a batch of this from the last surviving mint from my garden, which wasn’t in the best shape (there’s an inch of snow on the ground, and the leaves are mostly turning black). Can you come up with a better version for winter? How about less citrus, and replacing the mint with cardamom or cloves?
This recipe illustrates a few techniques beyond the basics: zesting a lemon and incorporating zest with the dry ingredients (hint: a fine Microplane is probably the most efficient tool for this); incorporating fresh citrus juice (strain, and add after the cream and eggs have been incorporated, to keep the milk proteins from curdling); and herbal infusion (done at low temperature—important with mint, to protect its delicate flavor) and the addition of essential oil, to compensate for the difficulty of infusing mint’s full flavor.
[edited 6-2018. Reduced mint essential oil from 0.5g to 0.1g. For reasons that will be obvious to anyone who tries it either way]
340g whole milk (3.3% fat)*
12g invert syrup
85g granulated sugar
55g nonfat dry milk*
4g (aprox.) lemon zest (from 1 small lemon)
1g salt
0.8g locust bean gum. increase to 1.0g if you get icy textures (tested with TIC Gums POR/A, soluble at 74°C)
0.4g guar gum
0.2g lambda carrageenan
2 large egg yolks (36g)
360g heavy cream (36% fat)*
24g (aprox) lemon juice (from 1 small lemon)
65g bourbon (43% alcohol = 28g)
2g Angostura bitters
12g very fresh mint leaves
*Use the best quality milk and cream you can get. Avoid anything ultrapasteurized. Low-temperature pasteurized is ideal. Homogenized products will give best texture. Avoid cream with added stabilizer (unknown variables). Dry milk should be 100% skim milk solids, processed without high heat. There should be no off odors either when it’s dry or when it’s mixed. Store sealed in freezer.
Next post: introduction to Flavor



HI Anon, thank you for the ice cream spreadsheet. It looks excellent. I've played with it a bit. My Excel skills aren't great, so I've been putting off editing it to match my own workflow and the way I think about recipes.
Why are you using maltodextrin instead of milk solids?
Thank you!
Thank you for this Booze Flavor Ice Cream. I will try your recipe.
– Gustavo Woltmann
Gracias! Impresionante!
Hi Paul,
Great post as always.
I hope you found the spreadsheet somewhat useful.
I really agree with the sentiment that a lot of the delicate ice creams should be less sweet. For alcoholic ice cream, I do pretty much the same thing (more stabilizer and no dextrose) but I also used maltodextrin for the contribution solids in both alcoholic sorbets and ice creams when the liquor is not reduced. It is barely considered sweet and does not affect the freezing point as much which makes it really useful for things like creme de menthe for example or Tia Maria which might have high sugar and alcohol by default.
Very enlightening post. Thank you. Elsewhere on your blog you mention a browned butter cognac if cream recipe. Would you mind posting that?
Hi Jesse, that’s a pretty old recipe and isn’t nearly as good as our current work, so I’m hesitant to publish it. Shoot me an email and I’ll send it to you. You can easily adapt the brown butter and cognac elements to other formulas.
Thanks. I am interested in gathering info from your recipes even if they aren’t your best. Can you also tell me what your thoughts are regarding the use of maltodextrin to achieve a reasonable freezing point when a higher percentage of alcohol is desired (above 5%)?
I’ve built my own model that estimates FDP using PAC , PAC(sa), and solids. I’m wondering if the amount of alcohol has it’s own additional freezing point depression calculation beyond the PAC associated with the specific ingredient. If not, do you find a use in tracking the specific quantities of alcohol in a recipe? How do you adjust PAC as you reduce the alcohol in a given spirit?
I have similar questions on acidity — do you have any plans for a post about acidity?
Hi Jack, I’m not quite sure I understand the alcohol question. Alcohol depresses freezing point in a linear fashion just like salts and most other solutes. You can calculate its FPD based purely on concentration and its molecular weight. Sugars are different; at higher concentrations, their FPD is non-linear, so the curves need to be extrapolated from experimental data if you want reasonably accurate results.
Adjusting PAC as you reduce alcohol is easy; just push the sugars in the direction of a normal recipe. The more challenging thing is to preserve the PAC as you increase alcohol. You have to reduce other ingredients that have a high PAC, especially dextrose and other low molecular weight sugars. Alcohol has so much freezing point depression that there’s a limit to what’s possible. At a certain point you have to decide if the ice cream will be boozy and soft, or weaker flavored and firm.
I’m still working on acidity. There’s a chance that acidity can have a thickening effect on the dairy, or in some cases can effect the emulsifying properties of denatured milk proteins. It definitely affects tartness. Right now I use the acidity value to help predict if I’ll need to add any lemon juice or citric acid to fruit ice creams. For example, if you’re making ice cream with fairly tart berries, you probably don’t want to do this. I made strawberry ice cream once (with added lemon) that tasted like frozen yogurt. Now I have some numbers to warn against this.
How hight of a concentration of sugars until the non-linearity becomes a practical issue in estimating FPD using a standard ΔTf = iKfm equation? I’ve seen some graphs that look fairly linear up till 15%-20% sugar. I can’t imagine going sweeter in any recipe. Do you think a linear assumption of sugars is fair for practical purposes (maybe not the theoretical). I imagine with that level of precision you’d need not only a polynomial regression for sugars, but a unique one for each type of sugar — as the non-linearity of sugar FPD is not going to be the same for each sugar. And at that point the entire idea of a PAC value becomes irrelevant.
You have to look at the process of fractional freezing. As ice crystals form, they form as pure water ice. Which means that all the solutes get forced out into the serum surrounding the crystal. They will then have a higher solids concentration, and so higher freezing point depression. Now the temperature needs to dip lower for more water to freeze. This concentrates the serum more, and the process continues.
Because of this process, every ice cream formula goes beyond 20% sugar concentration (in the unfrozen portion) when it freezes hard.
The freezing curve isn’t THAT far from linear. I haven’t gone through the trouble of making a spreadsheet to see how different the results would be in practical terms. Here’s a graph from dairyscience.ort: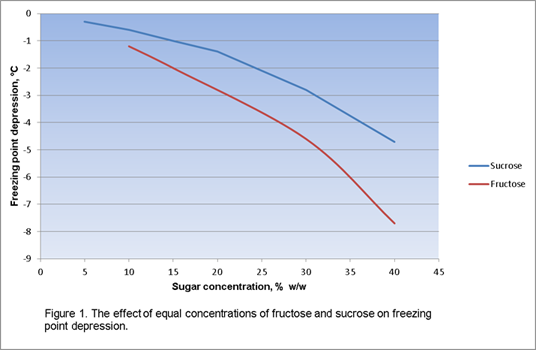
We calculate sugar solution freezing point with two different models. One is a simple predictive equation that’s used in many kinds of food products. We’ve tweaked one of the constants to bring the results close to experimental data. This is a quick method that lets you type in a serving temperature and get an ice fraction estimate. The second is a full-on regression using a polynomial curve that’s been fitted to the experimental results for sugar solutions. This is good for producing a graph, and is probably a bit more accurate.
I believe the non-linearities of sucrose, fructose, and glucose are very close to each other, if not identical, once you normalize for molecular weight.
Thanks for the reply. I guess I am wondering if there is a way to convert the FDP of alcohol into a sucrose-equivalency, as is often done with salts (ie PAC(sa)) — some sort of PAC(alc)?
Sure. The molecular weight of sucrose is 7.4 times that of ethanol. So, not accounting for the non-linearities of sucrose, you’d expect a gram of ethanol to have the FPD of 7.4g of sucrose.
You’re generally right that PAC is of limited use, if you’re trying to be precise. We calculate PAC just so people like pastry chefs and gelato makers who are trained in PAC will have some idea what we’re talking about. But depending on the formulas you’re dealing with, comparing the PAC of one to the PAC another may well be an apples/oranges situation … the numbers won’t accurately predict results.
We’re trying to figure out how to put a simplified version of our software online, so everyone can use the more accurate models that we use. It’s not quite fair right now that we explain ice cream in terms of general scientific principles, when the application of these principles in real life is actually very complex.
hello,
Could you explain us how to calculate pac & pod according to the degree of alcohol and brix of liquor we add ?
thank you